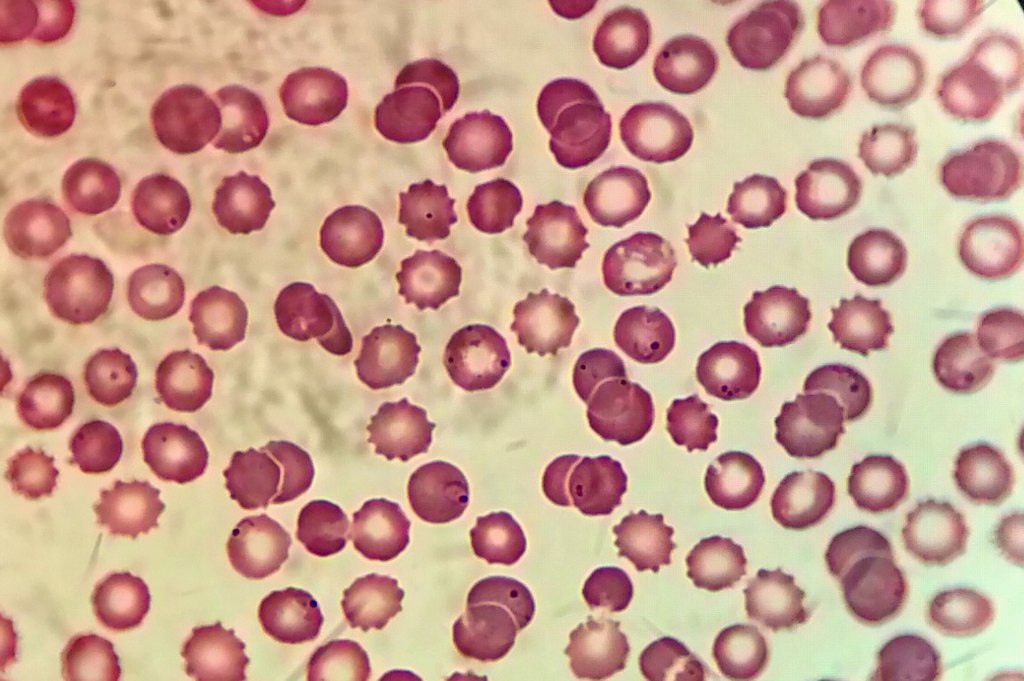
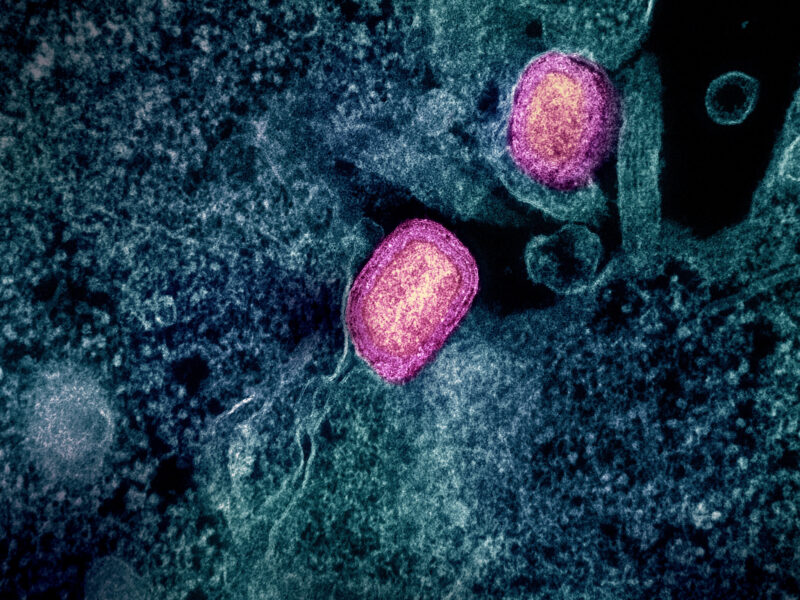
Image courtesy of Juan Carlos Fonseca Mata; CC-BY-SA.
Malaria is a class of parasites (P. falciparum, P. vivax, P. malariae, P. ovale, and P. knowlesi.) that causes more than 240 million cases of malaria infection each year, 95% of which are concentrated in Africa1,2. Malaria that infects humans arose from parasites that infects gorillas and apes3. Most infections are acquired after an individual is bitten by an infected female Anopheles mosquito. These mosquitoes acquire the parasites when they feed on an infected person and then are able to transmit that infection to a new host when they feed again. Since malaria is a blood borne parasite it is also able to be transmitted via blood transfusions, organ transplants, shared needles, and from mother to baby during childbirth1.
Malaria infection spreads systemically, encapsulated in the host’s red blood cells. It can take 10 days for symptoms to present, but once the parasite has invaded the blood cells, individuals can exhibit fever, chills, headache, muscle ache, tiredness, nausea, vomiting and diarrhea. As red blood cells are destroyed by the parasite, a person may present with anemia and jaundice, and if the infection is not treated kidney failure, seizures, confusion, coma and death can result4.
A common treatment for malaria was an oral medication called chloroquine. Chloroquine increases the pH of the parasite’s food vacuole which causes an accumulation of free hematin and toxins which eventually kills the parasite5. However, this form of treatment became increasingly inefficacious as the parasite developed resistance6. In its stead a new first line of treatment was developed, called artemisinin-based combination therapies (ACTs)7. ACTs are believed to work via a two-pronged mechanism. Artemisinin is activated by heme-iron which builds up within the parasite and then catalyzes a cleavage reaction. Once that reaction has occurred the resulting free radical intermediate may end up poisoning the parasite8. By using a combination of medications, any parasites that were not cleared by the artemisinin are dealt with by the partner drugs.
Despite this method’s initial success, in 2009 researchers began to see signs that resistance was developing. According to scientists, the artemisinin was failing to clear the parasite quickly or effectively so the partner drugs were being left to pick up its slack. With these partner drugs being exposed to higher parasite loads, the parasites were developing resistance to them9. According to an article written by Nicholas White in Manson’s Tropical Infectious Diseases, it is believed that antimalarial resistance is the result of spontaneous chromosomal point mutations as well as drug selection pressure10. Though this resistance began as an isolated incident along the Thailand-Cambodia border it has recently been identified in Africa, more specifically in Uganda and Rwanda. To add to concern, scientists have discovered that the genetic mutations that are leading to resistance have evolved independently of each other. This means that these mutations are unique and unrelated but have caused the same outcome9.
Currently, the primary course of action rests heavily upon the shoulders of community-based public health workers who are charged with surveillance. However, these communities are under-resourced and current methods of monitoring are woefully ill equipped, which has led to global concern9. A small bright spot has emerged recently as the WHO approved the first malaria vaccine. This vaccine is currently in the pilot stages of rollout in 12 African countries11. Though this development is promising for the prevention of malaria infection, especially for children who are most at risk, it does little to help those who have already contracted the parasite. In order for progress to be made to mitigate morbidity and mortality it is paramount that international funding continue to bolster monitoring, treatment and vaccination efforts.
References:
- CDC – Malaria – About Malaria – FAQs. www.cdc.gov/malaria/about/faqs.html#:~:text=The%20Disease,-What%20is%20malaria&text=Malaria%20is%20a%20serious%20and,%2C%20and%20flu%2Dlike%20illness.
- Health Alert Network (HAN) – 00494 | Locally Acquired Malaria Cases Identified in the United States. emergency.cdc.gov/han/2023/han00494.asp#:~:text=Worldwide%2C%20more%20than%20240%20million,Saharan%20Africa%20and%20South%20Asia.
- Loy, Dorothy E., et al. “Out of Africa: Origins and Evolution of the Human Malaria Parasites Plasmodium Falciparum and Plasmodium Vivax.” International Journal for Parasitology, vol. 47, no. 2–3, Elsevier BV, Feb. 2017, pp. 87–97, doi:10.1016/j.ijpara.2016.05.008.
- CDC – Malaria – Diagnosis and Treatment (United States) – Treatment (U.S.) – Guidelines for Clinicians. www.cdc.gov/malaria/diagnosis_treatment/clinicians1.html#:~:text=The%20preferred%20antimalarial%20for%20interim,not%20adequate%20for%20interim%20treatment.
- Zhou, Wenmin, et al. “Chloroquine Against Malaria, Cancers and Viral Diseases.” Drug Discovery Today, vol. 25, no. 11, Elsevier BV, Nov. 2020, pp. 2012–22, doi:10.1016/j.drudis.2020.09.010.
- CDC – Malaria – Malaria Worldwide – How Can Malaria Cases and Deaths Be Reduced? – Drug Resistance in the Malaria Endemic World. www.cdc.gov/malaria/malaria_worldwide/reduction/drug_resistance.html.
- World Health Organization and Global Malaria Programme. Artemisinin Resistance and Artemisinin-based Combination Therapy Efficacy. Dec. 2018, www.who.int/docs/default-source/documents/publications/gmp/who-cds-gmp-2018-26-eng.pdf.
- “Artemisinin Antimalarials: Mechanisms of Action and Resistance.” PubMed, 1998, pubmed.ncbi.nlm.nih.gov/10212891/#:~:text=Artemisinin%20is%20believed%20to%20act,essential%20malarial%20protein(s).
- Pawar, Pratik. “Drug-resistant Malaria Is Emerging in Africa. Doctors Are Worried — yet Hopeful.” NPR, 6 Feb. 2022, www.npr.org/sections/goatsandsoda/2022/02/06/1077953012/drug-resistant-malaria-is-emerging-in-africa-doctors-are-worried-yet-hopeful.
- White, Nicholas J. “Malaria.” Elsevier eBooks, 2014, pp. 532-600.e1, doi:10.1016/b978-0-7020-5101-2.00044-3.
- Malaria Vaccine Implementation Programme. 5 July 2023, www.who.int/initiatives/malaria-vaccine-implementation-programme