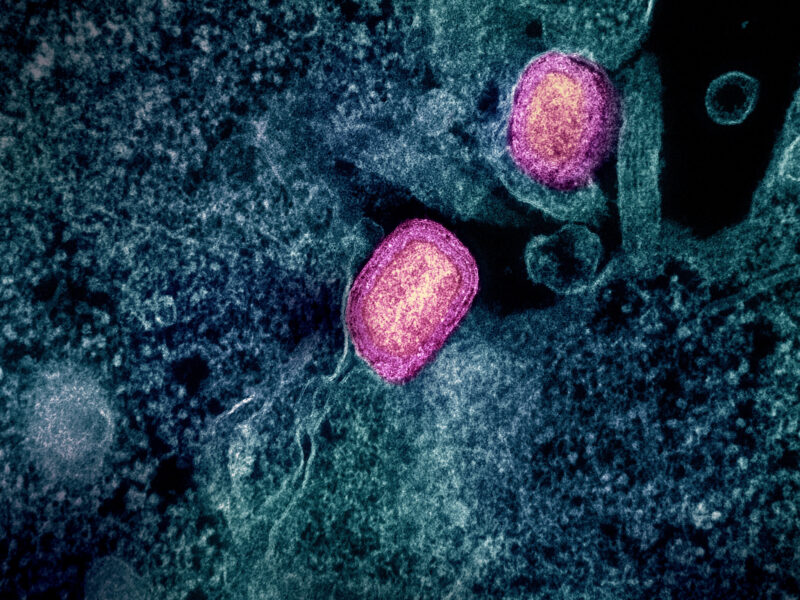
In a recent study published online by Immunity this January, researchers have been able to successfully isolate four antibodies against human immunodeficiency virus (HIV) from HIV vaccines after a phase III trial in Thailand. The trial, called RV144, involved approximately 16,000 volunteers and was conducted by researchers from the United States Army, the National Institutes of Health, the Thai Ministry of Public Health and two vaccine companies.
The trial itself was completed in 2009. The group from the trial that received the HIV vaccine had an infection rate 31.2 percent lower than the group who received the placebo. Later in 2011, researchers from Duke University used data from the trial to conduct a new analysis using Bayesian statistics and revised the chance of being ineffective from 4% to above 22%.
The newly found antibodies are considered to neutralize HIV, and recognize and mediate the killing of infected cells. This is a significant finding because these new antibodies may provide clues to develop the HIV vaccine.
We have been waiting for an HIV vaccine. There are, however, numerous obstacles, and progress is seemingly slow. Further, HIV offers challenges to vaccine production. Vaccines are made by either taking a part of a virus or bacteria, weakening a virus, or inactivating a virus, and giving it to the human body.
Because the pathogen is either weakened, inactive, or incomplete, it does not produce an illness. However, the immune system recognizes the pathogen, and develops to protect against it in the future. This isn’t really an option with HIV because the virus mutates rapidly and the possibility remains that the attenuated virus will transform again to be infectious. Further, since there are currently two types of virus (HIV-1 and HIV-2) and more than 3 subgroups with at least 9 subtypes for HIV-1 due to rapid mutation, it is difficult to develop a vaccine that works for all strains.
Next, and this does not apply to only HIV vaccine development, it is very difficult to conduct even pre-clinical trials in some countries. Experimental studies usually involve chimpanzees, but due to financial and ethical reasons, many vaccine candidates suffered setbacks at pre-clinical stage, which involves in-vitro and animal testing.
Third, the cost of development of the HIV vaccine is enormous, yet the vaccine needs to be made affordable to people with a range of socioeconomic statuses. Since the vaccine has to be provided inexpensively for many vulnerable populations, the cost performance is not attractive to sponsors and companies. Despite obstacles, researchers, organizations and companies worldwide unite efforts and break through the barriers to continue to develop effective HIV vaccines. According to International AIDS Vaccine Initiative (IAVI),1 many clinical trials are ongoing or completed worldwide.
Vaccine trials occur in four different phases. In the pre-clinical trials phase, vaccines are tested on animals. In phase I, the safety of the vaccine is tested. Researchers give a small number of people the vaccine and look for side effects. In phase II, researchers enroll a larger population and look for the strength of the immune response. Researchers consider this the “proof of concept” phase; can the immune response defeat the pathogen? In the final phase, phase III,, thousands of people may be enrolled. Researchers gather final evidence to determine if the vaccine is safe, efficient, and acceptable to the FDA. Currently, there are only three trials that have completed phase III.
The analysis of the RV144 trial is still ongoing and further studies suggest optimistic outcomes for HIV vaccine development. And this isn’t the only good news. A recent study from Johns Hopkins University School of Medicine reported that by removing cholesterol from the outer membrane of HIV, one can disable the virus from attacking the immune system.
Further, a new HIV vaccine (SAV001-H), developed in Canada, was approved by FDA last March to proceed to human trials. The vaccine demonstrated boosting immune effect after Phase I. So far, no adverse effect is observed. This vaccine contains killed HIV virus, which is similar to some influenza, polio, hepatitis A and rabies vaccines. The phase II trial has yet to be conducted.